 This difficulty was solved by Niels Bohr (1913), who applied the quantum theory developed by Max Planck and Albert Einstein to the problem of atomic structure. Thus, they should lose their energy and spiral into the nucleus. According to classical theory, as the electrons orbit about the nucleus, they are continuously being accelerated (see acceleration), and all accelerated charges radiate electromagnetic energy. There was one serious obstacle to acceptance of the nuclear atom, however. Using alpha particles emitted by radioactive atoms, he showed that the atom consists of a central, positively charged core, the nucleus, and negatively charged particles called electrons that orbit the nucleus. In 1911, Ernest Rutherford developed the first coherent explanation of the structure of an atom. it was generally accepted that matter is composed of atoms that combine to form molecules. Meyer, which arranged atoms of different elements in order of increasing atomic weight so that elements with similar chemical properties fell into groups. A major development was the periodic table, devised simultaneously by Dmitri Mendeleev and J. The best evidence for his theory was the experimentally verified law of simple multiple proportions, which gives a relation between the weights of two elements that combine to form different compounds.Įvidence for Dalton's theory also came from Michael Faraday's law of electrolysis. He stated that atoms of the elements unite chemically in simple numerical ratios to form compounds. He held that all the atoms of an element are of exactly the same size and weight (see atomic weight) and are in these two respects unlike the atoms of any other element. 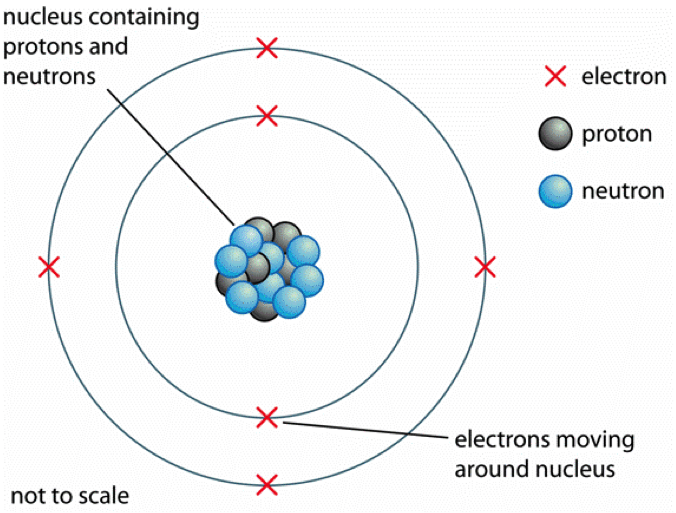
Modern atomic theory begins with the work of John Dalton, published in 1808. following work on the nature and behavior of gases (see gas laws). Interest in the atomic theory was revived during the 18th cent. However, Aristotle did not accept the theory, and it was ignored for many centuries. 
BC by the Greek philosophers Leucippus and Democritus and was adopted by the Roman Lucretius. The atomic theory, which holds that matter is composed of tiny, indivisible particles in constant motion, was proposed in the 5th cent. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
